-
抗体試薬
- フローサイトメトリー用試薬
-
ウェスタンブロッティング抗体試薬
- イムノアッセイ試薬
-
シングルセル試薬
- BD® AbSeq Assay | シングルセル試薬
- BD Rhapsody™ Accessory Kits | シングルセル試薬
- BD® Single-Cell Multiplexing Kit | シングルセル試薬
- BD Rhapsody™ Targeted mRNA Kits | シングルセル試薬
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit | シングルセル試薬
- BD Rhapsody™ TCR/BCR Profiling Assays (VDJ Assays) | シングルセル試薬
- BD® OMICS-Guard Sample Preservation Buffer
- BD Rhapsody™ ATAC-Seq Assays
-
細胞機能評価のための試薬
-
顕微鏡・イメージング用試薬
-
細胞調製・分離試薬
-
- BD® AbSeq Assay | シングルセル試薬
- BD Rhapsody™ Accessory Kits | シングルセル試薬
- BD® Single-Cell Multiplexing Kit | シングルセル試薬
- BD Rhapsody™ Targeted mRNA Kits | シングルセル試薬
- BD Rhapsody™ Whole Transcriptome Analysis (WTA) Amplification Kit | シングルセル試薬
- BD Rhapsody™ TCR/BCR Profiling Assays (VDJ Assays) | シングルセル試薬
- BD® OMICS-Guard Sample Preservation Buffer
- BD Rhapsody™ ATAC-Seq Assays
- Japan (Japanese)
-
Change country/language
Old Browser
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?


.png)

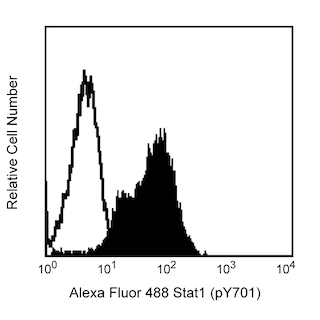
Analysis of Btk (pY551) in activated human B lymphoma cells. Ramos cells (ATCC CRL-1596) were serum starved overnight and then either stimulated with 5 mM hydrogen peroxide for 15 minutes (shaded histogram) or unstimulated (open histogram). The cells were fixed (BD Cytofix™ buffer, Cat. No. 554655) for 10 minutes, then permeabilized (BD Phosflow™ Perm Buffer III, Cat. No. 558050) on ice for at least 30 minutes, and then stained with Alexa Fluor® 488 anti-Btk (pY551)/Itk (pY511). Flow cytometry was performed on a BD FACSCalibur™ flow cytometry system.
.png)

BD Phosflow™ Alexa Fluor® 488 Mouse Anti-Btk (pY551)/Itk (pY511)
.png)
Regulatory Statusの凡例
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation and Storage
Product Notices
- This reagent has been pre-diluted for use at the recommended Volume per Test. We typically use 1 × 10^6 cells in a 100-µl experimental sample (a test).
- For fluorochrome spectra and suitable instrument settings, please refer to our Multicolor Flow Cytometry web page at www.bdbiosciences.com/colors.
- The Alexa Fluor®, Pacific Blue™, and Cascade Blue® dye antibody conjugates in this product are sold under license from Molecular Probes, Inc. for research use only, excluding use in combination with microarrays, or as analyte specific reagents. The Alexa Fluor® dyes (except for Alexa Fluor® 430), Pacific Blue™ dye, and Cascade Blue® dye are covered by pending and issued patents.
- Alexa Fluor® 488 fluorochrome emission is collected at the same instrument settings as for fluorescein isothiocyanate (FITC).
- Alexa Fluor® is a registered trademark of Molecular Probes, Inc., Eugene, OR.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- Source of all serum proteins is from USDA inspected abattoirs located in the United States.
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
Bruton's tyrosine kinase (Btk) is a nonreceptor tyrosine kinase whose function is critical for proper B cell development and signaling. The activity of Btk is regulated by Src mediated phosphorylation of the kinase domain at tyrosine 551 (Y551). This event induces Btk kinase activity and subsequent autophosphorylation at Y223. Phosphorylated Btk then associates with the cell membrane via the interaction of the PH domain with phosphatidylinositol 3, 4, 5-triphosphate.
The Tec family kinase Itk plays a critical role in signal transduction downstream of the T cell antigen receptor and has been implicated in the activation of phospholipase C-γ1 (PLCγ1), a key regulator of calcium mobilization and extracellular signal-regulated kinase (ERK) activation. Itk is regulated by an activating transphosphorylation event in which Y511 in the kinase domain is phosphorylated by Lck.
The 24a/BTK (Y551) monoclonal antibody recognizes the Y551-phosphorylated form of human Btk and the Y511 phosphorylated form of human Itk.
Development References (3)
-
Mahajan S, Fargnoli J, Burkhardt AL, Kut SA, Saouaf SJ, Bolen JB. Src family protein tyrosine kinases induce autoactivation of Bruton's tyrosine kinase. Mol Cell Biol. 1995; 15:5304-5311. (Biology). View Reference
-
Rawlings DJ, Scharenberg AM, Park H, et al. Activation of BTK by a phosphorylation mechanism initiated by SRC family kinases. Science. 1996; 271:822-825. (Biology). View Reference
-
Wilcox HM, Berg LJ.. Itk phosphorylation sites are required for functional activity in primary T cells. J Biol Chem. 2003; 278:37112-37121. (Biology).
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Report a Site Issue
This form is intended to help us improve our website experience. For other support, please visit our Contact Us page.