Old Browser
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?
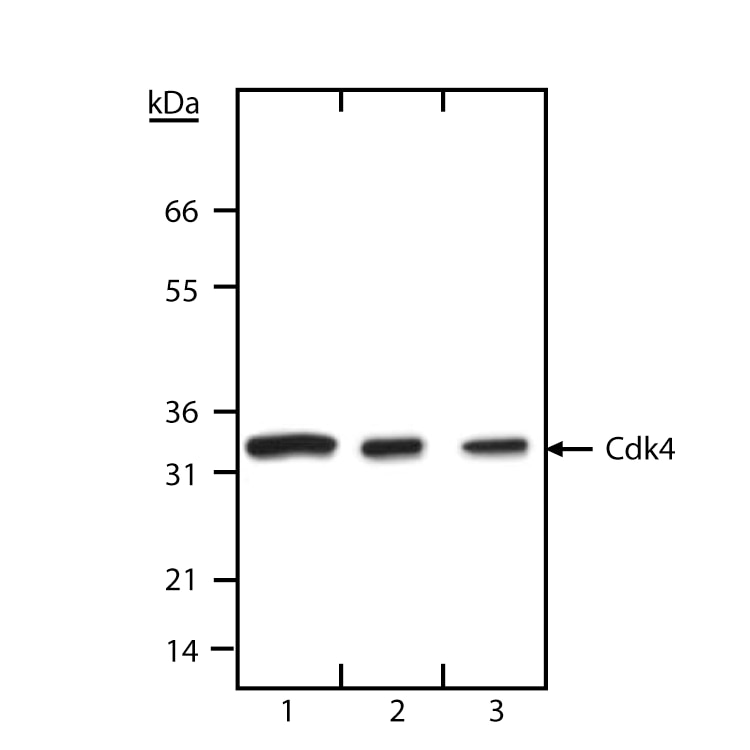




Western blot analysis of Cdk4. 293 cell lysates were probed with 5 µg/ml (lane 1), 2 µg/ml (lane 2) or 0.5 µg/ml (lane 3) of anti-Cdk4 (clone DCS-35, Cat. No. 68941A). The antibody identifies Cdk4 at ~33 kDa.

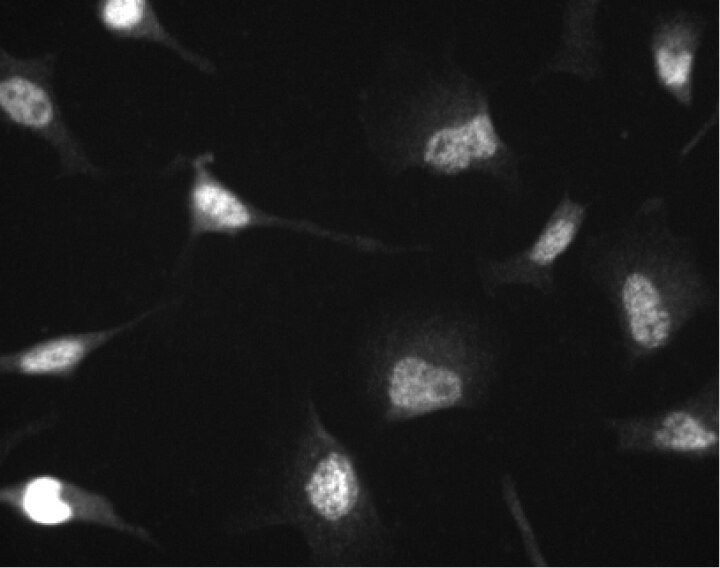
Immunofluorescent staining of HeLa (ATCC CCL-2) cells. Cells were seeded in a 96 well imaging plate (Cat. No. 353219) at ~10 000 cells per well. After overnight incubation, cells were stained using the alcohol perm protocol and the anti-Cdk4 antibody. The second step reagent was Alexa Fluor® 555 (Invitrogen). The image was taken on a BD Pathway™ 855 Bioimager system using a 20x objective. This antibody also stained A549 (ATCC CCL-185) and U-2 OS (ATCC HTB-96) cells and worked with both the Triton™ X-100 and alcohol perm protocols (see Recommended Assay Procedure).


BD Pharmingen™ Purified Anti-Human Cdk4 (N-Term)

BD Pharmingen™ Purified Anti-Human Cdk4 (N-Term)

Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
Recommended Assay Procedures
Bioimaging
1. Seed the cells in appropriate culture medium at ~10,000 cells per well in a BD Falcon™ 96-well Imaging Plate (Cat. No. 353219) and culture overnight.
2. Remove the culture medium from the wells, and fix the cells by adding 100 μl of BD Cytofix™ Fixation Buffer (Cat. No. 554655) to each well. Incubate for 10 minutes at room temperature (RT).
3. Remove the fixative from the wells, and permeabilize the cells using either BD Perm Buffer III, 90% methanol, or Triton™ X-100:
a. Add 100 μl of -20°C 90% methanol or Perm Buffer III (Cat. No. 558050) to each well and incubate for 5 minutes at RT.
OR
b. Add 100 μl of 0.1% Triton™ X-100 to each well and incubate for 5 minutes at RT.
4. Remove the permeabilization buffer, and wash the wells twice with 100 μl of 1× PBS.
5. Remove the PBS, and block the cells by adding 100 μl of BD Pharmingen™ Stain Buffer (FBS) (Cat. No. 554656) to each well. Incubate for 30 minutes at RT.
6. Remove the blocking buffer and add 50 μl of the optimally titrated primary antibody (diluted in Stain Buffer) to each well, and incubate for 1 hour at RT.
7. Remove the primary antibody, and wash the wells three times with 100 μl of 1× PBS.
8. Remove the PBS, and add the second step reagent at its optimally titrated concentration in 50 μl to each well, and incubate in the dark for 1 hour at RT.
9. Remove the second step reagent, and wash the wells three times with 100 μl of 1× PBS.
10. Remove the PBS, and counter-stain the nuclei by adding 200 μl per well of 2 μg/ml Hoechst 33342 (e.g., Sigma-Aldrich Cat. No. B2261) in 1× PBS to each well at least 15 minutes before imaging.
11. View and analyze the cells on an appropriate imaging instrument.
Bioimaging: For more detailed information please refer to http://www.bdbiosciences.com/support/resources/protocols/ceritifed_reagents.jsp
Western blot: For more detailed information please refer to http://www.bdbiosciences.com/pharmingen/protocols/Western_Blotting.shtml
Product Notices
- Since applications vary, each investigator should titrate the reagent to obtain optimal results.
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
- This antibody has been developed and certified for the bioimaging application. However, a routine bioimaging test is not performed on every lot. Researchers are encouraged to titrate the reagent for optimal performance.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- Triton is a trademark of the Dow Chemical Company.
Companion Products



Cyclins, cyclin-dependent kinases (Cdks), and cyclin-dependent kinase inhibitors (CdkIs) are essential for cell-cycle control in eukaryotes (reviewed in 1). Cyclins, regulatory subunits, bind to cyclin-dependent kinases (Cdks), catalytic subunits, to form active cyclin-Cdk complexes. Cdk subunits by themselves are inactive and binding to a cyclin is required for their activity. Cyclins A, B1, D and E undergo periodic synthesis and degradation, thereby providing a mechanism to regulate Cdk activity throughout the cell cycle. Additionally, Cdk activity is further regulated by activating and inhibitory phosphorylations, and small proteins (p15, p16, p18, p19, p21 and p27), called CdkIs, that bind to cyclins, Cdks, or cyclin-Cdk complexes. Cdk4 was originally called PSK-J3,2 and following demonstration of its association with D-type cyclins, became known as Cdk4.2 D-type cyclins also associate with Cdks 2 and 5, although Cdk4 appears to be the most abundant partner. The D-type cyclins (D1, D2, and D3) are expressed in response to growth factors or mitogens, and rapidly degrade when mitogens are withdrawn. D cyclins appear to promote G0 to G1 transitions and the rate of G1 progression. For example, cyclin D-Cdk4 and cyclin D-Cdk6 complexes phosphorylate the retinoblastoma protein (Rb) which removes the G1 phase block caused by underphosphorylated Rb. Cdk4 has a molecular weight of ~33 kD.
Development References (3)
-
Depoortere F, Van Keymeulen A, Lukas J, et al. A requirement for cyclin D3-cyclin-dependent kinase (cdk)-4 assembly in the cyclic adenosine monophosphate-dependent proliferation of thyrocytes. J Cell Biol. 1998; 140(6):1427-1439. (Immunogen). View Reference
-
Johnson DG, Walker CL. Cyclins and cell cycle checkpoints. Annu Rev Pharmacol Toxicol. 1999; 39:295-312. (Biology). View Reference
-
Matsushime H, Ewen ME, Strom DK, et al. Identification and properties of an atypical catalytic subunit (p34PSK-J3/cdk4) for mammalian D type G1 cyclins. Cell. 1992; 71(2):323-334. (Biology). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Refer to manufacturer's instructions for use and related User Manuals and Technical Data Sheets before using this product as described.
Comparisons, where applicable, are made against older BD technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
Report a Site Issue
This form is intended to help us improve our website experience. For other support, please visit our Contact Us page.