Old Browser
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?




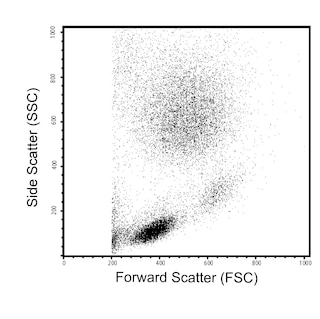
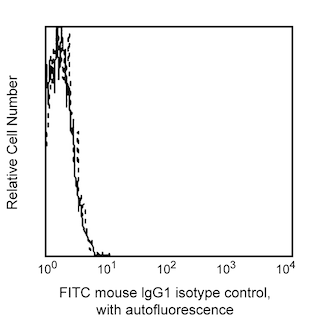
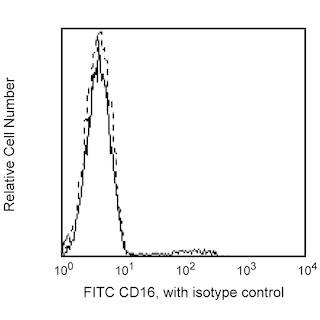
Flow cytometric analysis of CD16 expression on Rhesus macaque peripheral blood cells. Peripheral blood cells from Rhesus macaque were stained with FITC Mouse Anti-Human CD16 antibody (Cat. No. 555406/556618/560996; solid line histogram) or FITC Mouse IgG1, κ Isotype Control (Cat. No. 555748; dashed line histogram). The erythrocytes were lysed with BD Pharm Lyse™ Lysing Buffer (Cat. No. 555899). The fluorescence histograms were derived from gated events with the forward and side light-scatter characteristics of viable lymphocytes. Flow cytometry was performed using a BD FACScan™ Flow Cytometer System.


BD Pharmingen™ FITC Mouse Anti-Human CD16

Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
Recommended Assay Procedures
BD™ CompBeads can be used as surrogates to assess fluorescence spillover (Compensation). When fluorochrome conjugated antibodies are bound to CompBeads, they have spectral properties very similar to cells. However, for some fluorochromes there can be small differences in spectral emissions compared to cells, resulting in spillover values that differ when compared to biological controls. It is strongly recommended that when using a reagent for the first time, users compare the spillover on cell and CompBead to ensure that BD Comp beads are appropriate for your specific cellular application.
Product Notices
- This reagent has been pre-diluted for use at the recommended Volume per Test. We typically use 1 × 10^6 cells in a 100-µl experimental sample (a test).
- An isotype control should be used at the same concentration as the antibody of interest.
- Source of all serum proteins is from USDA inspected abattoirs located in the United States.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- For fluorochrome spectra and suitable instrument settings, please refer to our Multicolor Flow Cytometry web page at www.bdbiosciences.com/colors.
- Species cross-reactivity detected in product development may not have been confirmed on every format and/or application.
- Please refer to http://regdocs.bd.com to access safety data sheets (SDS).
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
Companion Products



The 3G8 monoclonal antibody specifically recognizes CD16a and CD16b, low affinity receptors for the Fc region of IgG. CD16a is ~50-65 kDa type I transmembrane glycoprotein that is encoded by FCGR3A (Fc fragment of IgG receptor IIIa) which belongs to the immunoglobulin superfamily. CD16a is also known as Fc-gamma RIII-alpha (Fc-gamma RIIIa or FcγRIIIA) or FcRIIIa and is expressed on natural killer cells, activated monocytes, macrophages, γδ T cells, immature thymocytes, and mast cells. CD16a binds immune-complexed or aggregated IgG and associates with CD247/TCRζ in NK cells and FcεRIγ chains in phagocytes and mast cells to transduce intracellular signals. CD16a functions in antibody-dependent cellular cytotoxicity (ADCC) and other antibody-dependent responses including phagocytosis, cytokine production or mediator release. CD16b is a ~48 kDa glycophosyl-phosphatidylinositol (GPI)-linked form that is encoded by FCGR3B (Fc fragment of IgG receptor IIIb). CD16b is also known as Fc-gamma RIII-beta (Fc-gamma RIIIb or FcγRIIIB) or FcRIIIb and is expressed on neutrophils and activated eosinophils. The extracellular region of CD16b is highly homologous to CD16a. CD16b also serves as a receptor for the Fc region of IgG and can bind immune-complexed or aggregated IgG and may be involved in neutrophil adhesion.
The 3G8 antibody also crossreacts with a subset of peripheral blood lymphocytes and monocytes, but not granulocytes, of baboon, rhesus, and cynomolgus monkeys. Multicolor analysis reveals that the distribution on lymphocytes is similar to that found in human studies with the majority of CD16-positive lymphocytes being both CD3 and CD20 negative.
Development References (5)
-
Barclay NA, Brown MH, Birkeland ML, et al, ed. The Leukocyte Antigen FactsBook. San Diego, CA: Academic Press; 1997.
-
Fleit HB, Wright SD, Unkeless JC. Human neutrophil Fc gamma receptor distribution and structure. Proc Natl Acad Sci U S A. 1982; 79(10):3275-3279. (Biology). View Reference
-
Knapp W. W. Knapp .. et al., ed. Leucocyte typing IV : white cell differentiation antigens. Oxford New York: Oxford University Press; 1989:1-1182.
-
Stroncek DF, Skubitz KM, Plachta LB, et al. Alloimmune neonatal neutropenia due to an antibody to the neutrophil Fc-gamma receptor III with maternal deficiency of CD16 antigen. Blood. 1991; 77(7):1572-1580. (Biology). View Reference
-
Wirthmueller U, Kurosaki T, Murakami MS, Ravetch JV. Signal transduction by Fc gamma RIII (CD16) is mediated through the gamma chain. J Exp Med. 1992; 175(5):1381-1390. (Biology). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Refer to manufacturer's instructions for use and related User Manuals and Technical Data Sheets before using this product as described.
Comparisons, where applicable, are made against older BD technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
Report a Site Issue
This form is intended to help us improve our website experience. For other support, please visit our Contact Us page.