Old Browser
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?


Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
Recommended Assay Procedures
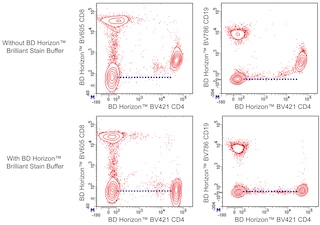
For optimal and reproducible results, BD Horizon Brilliant Stain Buffer should be used anytime two or more BD Horizon Brilliant dyes (including BD OptiBuild Brilliant reagents) are used in the same experiment. Fluorescent dye interactions may cause staining artifacts which may affect data interpretation. The BD Horizon Brilliant Stain Buffer was designed to minimize these interactions. More information can be found in the Technical Data Sheet of the BD Horizon Brilliant Stain Buffer (Cat. No. 563794).
Product Notices
- This antibody was developed for use in flow cytometry.
- The production process underwent stringent testing and validation to assure that it generates a high-quality conjugate with consistent performance and specific binding activity. However, verification testing has not been performed on all conjugate lots.
- Researchers should determine the optimal concentration of this reagent for their individual applications.
- An isotype control should be used at the same concentration as the antibody of interest.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- For fluorochrome spectra and suitable instrument settings, please refer to our Multicolor Flow Cytometry web page at www.bdbiosciences.com/colors.
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
- BD Horizon Brilliant Stain Buffer is covered by one or more of the following US patents: 8,110,673; 8,158,444; 8,575,303; 8,354,239.
- BD Horizon Brilliant Violet 786 is covered by one or more of the following US patents: 8,110,673; 8,158,444; 8,227,187; 8,455,613; 8,575,303; 8,354,239.
- Cy is a trademark of Amersham Biosciences Limited.
Companion Products
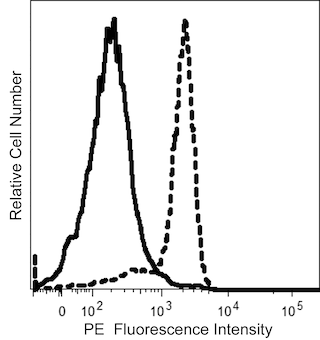



The 8C11 monoclonal antibody recognizes the extracellular domain of human N-Cadherin (CD325). Cadherins are a family of Ca2+ -dependent intercellular adhesion molecules that play a central role in controlling morphogenetic movements during development. Their function is regulated by association with the actin cytoskeleton by a complex of cytoplasmic proteins called the catenins (α, β, γ). Members of the cadherin family include P-cadherin , E-cadherin (uvomorulin), N-cadherin (neural cadherin), R-cadherin, cadherin 5, L-CAM, and EP-cadherin. N-cadherin mRNA is found at elevated levels in brain and heart and at a much lower level in liver. Mechanisms such as mRNA expression, cytokine modulation, and protease-mediated turnover modulate N-cadherin protein levels during development. In addition, N-cadherin function is indirectly regulated by endogenous kinases and phosphatases. Tyrosine phosphorylation of β-catenin complexed with N-cadherin results in dissociation of N-cadherin from actin. However, N-cadherin also interacts with a PTP1B-like phosphatase that dephosphorylates β-catenin and promotes N-cadherin/actin association. Thus, N-cadherin is an integral adhesion molecule whose function is regulated by protein-protein interactions and phosphorylation/dephosphorylation events.
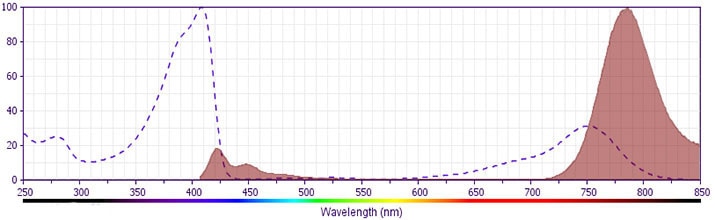
The antibody was conjugated to BD Horizon™ BV786 which is part of the BD Horizon Brilliant™ Violet family of dyes. This dye is a tandem fluorochrome of BD Horizon BV421 with an Ex Max of 405-nm and an acceptor dye with an Em Max at 786-nm. BD Horizon BV786 can be excited by the violet laser and detected in a filter used to detect Cy™7-like dyes (eg, 780/60-nm filter).
Development References (4)
-
Kim JB, Islam S, Kim YJ, et al. N-Cadherin extracellular repeat 4 mediates epithelial to mesenchymal transition and increased motility. J Cell Biol. 2000; 151(6):1193-1206. (Immunogen: Functional assay, Immunofluorescence, Inhibition, Western blot). View Reference
-
Knudsen KA, Soler AP, Johnson KR, Wheelock MJ. Interaction of alpha-actinin with the cadherin/catenin cell-cell adhesion complex via alpha-catenin. J Cell Biol. 1995; 130:66-77. (Biology). View Reference
-
Puch S, Armeanu S, Kibler C, et al. N-cadherin is developmentally regulated and functionally involved in early hematopoietic cell differentiation. J Cell Sci. 2001; 114(8):1567-1577. (Clone-specific: Flow cytometry). View Reference
-
Wein F, Pietsch L, Saffrich R, et al. N-Cadherin is expressed on human hematopoietic progenitor cells and mediates interaction with human mesenchymal stromal cells. Stem Cell Res. 2010; 4(2):129-139. (Clone-specific: Flow cytometry). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Refer to manufacturer's instructions for use and related User Manuals and Technical Data Sheets before using this product as described.
Comparisons, where applicable, are made against older BD technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
Report a Site Issue
This form is intended to help us improve our website experience. For other support, please visit our Contact Us page.