Old Browser
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?


.png)

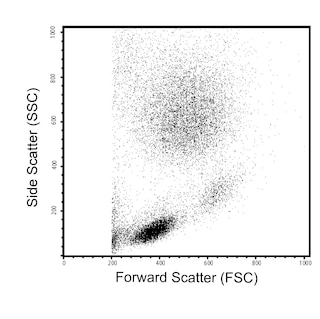
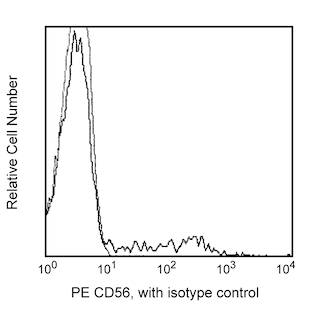
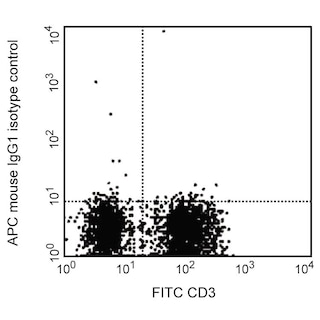
Multicolor flow cytometric analysis of GPR56 expression on human peripheral blood lymphocytes. Whole human blood was stained with PE Mouse Anti-Human CD56 antibody (Cat. No. 555516/561903) and either APC Mouse IgG1, κ Isotype Control (Cat. No. 554681; Left Plot) or APC Mouse Anti-Human GPR56 antibody (Cat. No. 567612/567613; Right Plot). Erythrocytes were lysed with BD FACS™ Lysing Solution (Cat. No. 349202). The bivariate pseudocolor density plots showing either GPR56 [or Ig isotype control] staining versus CD56 were derived from gated events with the forward and side light-scatter characteristics of intact lymphocytes. Flow cytometry and data analysis were performed using a BD LSRFortessa™ X-20 Cell Analyzer System and FlowJo® software.
.png)

BD Pharmingen™ APC Mouse Anti-Human GPR56
.png)
Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
Recommended Assay Procedures
BD® CompBeads can be used as surrogates to assess fluorescence spillover (Compensation). When fluorochrome conjugated antibodies are bound to BD® CompBeads, they have spectral properties very similar to cells. However, for some fluorochromes there can be small differences in spectral emissions compared to cells, resulting in spillover values that differ when compared to biological controls. It is strongly recommended that when using a reagent for the first time, users compare the spillover on cells and BD® CompBeads to ensure that BD® CompBeads are appropriate for your specific cellular application.
Product Notices
- This reagent has been pre-diluted for use at the recommended Volume per Test. We typically use 1 × 10^6 cells in a 100-µl experimental sample (a test).
- An isotype control should be used at the same concentration as the antibody of interest.
- Source of all serum proteins is from USDA inspected abattoirs located in the United States.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
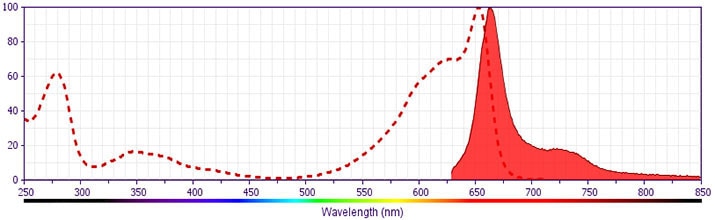
- This APC-conjugated reagent can be used in any flow cytometer equipped with a dye, HeNe, or red diode laser.
- For fluorochrome spectra and suitable instrument settings, please refer to our Multicolor Flow Cytometry web page at www.bdbiosciences.com/colors.
- Please refer to http://regdocs.bd.com to access safety data sheets (SDS).
- Species cross-reactivity detected in product development may not have been confirmed on every format and/or application.
Companion Products



The CG4.rMab is a recombinant monoclonal antibody that was derived from CG4 hybridoma cells. The CG4.rMab specifically binds to human G-protein coupled receptor 56 (GPR56) like the conventional CG4 antibody and performs like the CG4 antibody when used to stain cells and analyze them by flow cytometry. GPR56 is also known as adhesion G-protein coupled receptor G1 (ADGRG1), or TM7XN1. GPR56 is a ~15kDa G protein-coupled receptor encoded by ADGRG1 which belongs to the adhesion-GPCR family that comprises 33 members in human. The extracellular region contains a mucin-like domain followed by a membrane proximal GPCR-autoproteolysis inducing (GAIN) domain, seven transmembrane regions and a cytoplasmic tail. The constitutive self-cleavage at the proteolytic site gives rise to a membrane spanning (C-terminal fragment or CTF) and an extracellular (N-terminal fragment or NTF) subunit that remain noncovalently bound, leading to the expression of a heterodimeric receptor at the cell surface. GPR56 is widely expressed with the highest levels of messenger found in the brain, heart, and thyroid gland. Recently, GPR56 was found to be variably expressed on platelets, cytotoxic NK cells and T lymphocytes including CD4+, CD8+, and γδ T cells. It was shown that GPR56 functions as an inhibitory receptor on NK cells through interaction with CD81. While GPR56 NTF associates with Tissue transglutaminase 2 and Collagen III (α-1), GPR56 CTF can recruit Gα proteins leading to the activation of mTOR and RhoA signaling pathways. GPR56 has been implicated in cell-cell interactions, adhesion, migration, and regulation of cell proliferation and survival of various cell types. New evidence also shows a role of GPR56 in tumor progression. Recently, the CG4 antibody was found to activate GPR56 in melanoma cells leading to an increase of IL-6 secretion, in a CD9/CD81-dependent manner.
Development References (6)
-
Della Chiesa M, Falco M, Parolini S, et al. GPR56 as a novel marker identifying the CD56dull CD16+ NK cell subset both in blood stream and in inflamed peripheral tissues.. Int Immunol. 2010; 22(2):91-100. (Biology: Flow cytometry). View Reference
-
Liu M, Parker RM, Darby K, et al. GPR56, a novel secretin-like human G-protein-coupled receptor gene.. Genomics. 1999; 55(3):296-305. (Biology). View Reference
-
Pabst C, Bergeron A, Lavallée VP, et al. GPR56 identifies primary human acute myeloid leukemia cells with high repopulating potential in vivo.. Blood. 2016; 127(16):2018-27. (Clone-specific: Flow cytometry). View Reference
-
Peng YM, van de Garde MD, Cheng KF, et al. Specific expression of GPR56 by human cytotoxic lymphocytes.. J Leukoc Biol. 2011; 90(4):735-40. (Immunogen: Flow cytometry). View Reference
-
Piao X, Hill RS, Bodell A, et al. G protein-coupled receptor-dependent development of human frontal cortex.. Science. 2004; 303(5666):2033-6. (Biology). View Reference
-
Rao TN, Marks-Bluth J, Sullivan J, et al. High-level Gpr56 expression is dispensable for the maintenance and function of hematopoietic stem and progenitor cells in mice.. Stem Cell Res. 2015; 14(3):307-22. (Clone-specific: Flow cytometry). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Refer to manufacturer's instructions for use and related User Manuals and Technical Data Sheets before using this product as described.
Comparisons, where applicable, are made against older BD technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
Report a Site Issue
This form is intended to help us improve our website experience. For other support, please visit our Contact Us page.