Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?
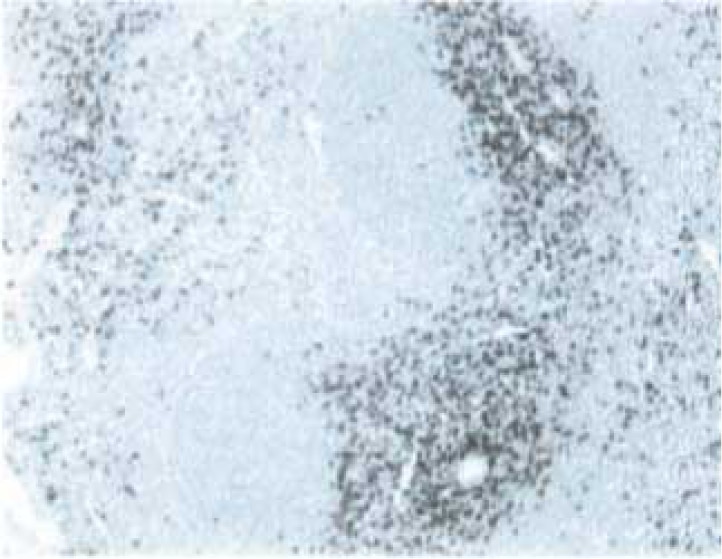



Immunohistochemical staining of Rat T lymphocytes. The paraffin-embedded section of normal rat spleen was stained with Purified Mouse Anti-Rat CD8a (Cat. No. 550298). CD8+ lymphocytes around the central arterioles of the white pulp are identified by the brown staining.


BD Pharmingen™ Purified Mouse Anti-Rat CD8a

Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
Recommended Assay Procedures
Immunohistochemistry: The OX-8 antibody is recommended to test for immunohistochemical staining of acetone-fixed frozen sections and paraffin sections. For paraffin sections no pretreatment is required. Tissues tested were rat spleen and thymus. The antibody stains the CD8 subset of T lymphocytes. The isotype control recommended for use with this antibody is purified mouse IgG1 (Cat. No. 550878). For optimal indirect immunohistochemical staining, the OX-8 antibody should be titrated (1:10 to 1:50 dilution) and visualized via a three-step staining procedure in combination with biotinylated polyclonal anti-mouse Ig (multiple adsorbed) (Cat. No. 550337) as the secondary antibody and Streptavidin-HRP (Cat. No. 550946) together with the DAB detection system (Cat. No. 550880). More conveniently, the anti-mouse Ig HRP detection kit (Cat. No. 551011) that contains the biotinylated secondary antibody, antibody diluent, streptavidin-HRP and DAB substrate can be used for staining.
Product Notices
- Since applications vary, each investigator should titrate the reagent to obtain optimal results.
- Source of all serum proteins is from USDA inspected abattoirs located in the United States.
- This antibody has been developed for the immunohistochemistry application. However, a routine immunohistochemistry test is not performed on every lot. Researchers are encouraged to titrate the reagent for optimal performance.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- Sodium azide is a reversible inhibitor of oxidative metabolism; therefore, antibody preparations containing this preservative agent must not be used in cell cultures nor injected into animals. Sodium azide may be removed by washing stained cells or plate-bound antibody or dialyzing soluble antibody in sodium azide-free buffer. Since endotoxin may also affect the results of functional studies, we recommend the NA/LE (No Azide/Low Endotoxin) antibody format, if available, for in vitro and in vivo use.
- An isotype control should be used at the same concentration as the antibody of interest.
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
- Please refer to http://regdocs.bd.com to access safety data sheets (SDS).
Companion Products



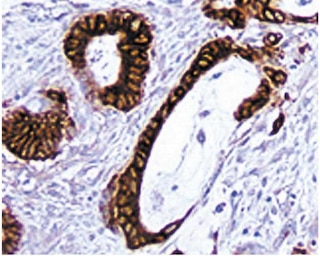

The OX-8 monoclonal antibody specifically binds to the hinge-like membrane-proximal domain of the 32 kDa α chain of the CD8 differentiation antigen. A truncated CD8 α' isoform has not been detected in the rat. The CD8 α and β chains (CD8a and CD8b, respectively) form a heterodimer on the surface of most thymocytes and a subpopulation of mature T lymphocytes (i.e., MHC class I-restricted T cells, including most T suppressor/cytotoxic cells). Intestinal intrapithelial lymphocytes, many CD8+ T cells of athymic rats, many activated CD4+ T cells, and most NK cells express CD8a without CD8b. It has been suggested that the expression of the CD8a/CD8b heterodimer is restricted to thymus-derived T lymphocytes. OX-8 antibody does not react with resting CD4+ T helper cells. CD8 is an antigen coreceptor on the T-cell surface which interacts with MHC class I molecules on antigen-presenting cells. It participates in T-cell activation through its association with the T-cell receptor complex and protein tyrosine kinase Ick. Macrophages have also been reported to express CD8 α and β chains, which are involved in signal transduction. Soluble OX-8 mAb partially blocks in vitro MLR and CTL activity.
Development References (11)
-
Barclay AN. The localization of populations of lymphocytes defined by monoclonal antibodies in rat lymphoid tissues. J Immunol. 1981; 42(4):593-600. (Clone-specific: Immunohistochemistry). View Reference
-
Bierer BE, Sleckman BP, Ratnofsky SE, Burakoff SJ. The biologic roles of CD2, CD4, and CD8 in T-cell activation. Annu Rev Immunol. 1989; 7:579-599. (Biology). View Reference
-
Brideau RJ, Carter PB, McMaster WR, Mason DW, Williams AF. Two subsets of rat T lymphocytes defined with monoclonal antibodies.. Eur J Immunol. 1980; 10:609-615. (Immunogen: Flow cytometry). View Reference
-
Classon BJ, Brown MH, Garnett D, et al. The hinge region of the CD8 alpha chain: structure, antigenicity, and utility in expression of immunoglobulin superfamily domains. Int Immunol. 1992; 4(2):215-225. (Clone-specific). View Reference
-
Janeway CA Jr. The T cell receptor as a multicomponent signalling machine: CD4/CD8 coreceptors and CD45 in T cell activation. Annu Rev Immunol. 1992; 10:645-674. (Biology). View Reference
-
Johnson P, Gagnon J, Barclay AN, Williams AF. Purification, chain separation and sequence of the MRC OX-8 antigen, a marker of rat cytotoxic T lymphocytes. EMBO J. 1985; 4(10):2539-2545. (Clone-specific: Immunoaffinity chromatography). View Reference
-
Mitnacht R, Bischof A, Torres-Nagel N, Hunig T. Opposite CD4/CD8 lineage decisions of CD4+8+ mouse and rat thymocytes to equivalent triggering signals: correlation with thymic expression of a truncated CD8 alpha chain in mice but not rats. J Immunol. 1998; 160(2):700-707. (Clone-specific: Immunoprecipitation, Western blot). View Reference
-
Stitz L, Sobbe M, Bilzer T. Preventive effects of early anti-CD4 or anti-CD8 treatment on Borna disease in rats. J Virol. 1992; 66(6):3316-3323. (Clone-specific: Blocking). View Reference
-
Thomas ML, Green JR. Molecular nature of the W3/25 and MRC OX-8 marker antigens for rat T lymphocytes: comparisons with mouse and human antigens. Eur J Immunol. 1983; 13(10):855-858. (Clone-specific: Immunoprecipitation). View Reference
-
Torres-Nagel N, Kraus E, Brown MH, et al. Differential thymus dependence of rat CD8 isoform expression.. Eur J Immunol. 1992; 22(11):2841-2848. (Clone-specific: Blocking, Immunoprecipitation, Western blot). View Reference
-
Wallgren AC, Karlsson-Parra A, Korsgren O. The main infiltrating cell in xenograft rejection is a CD4+ macrophage and not a T lymphocyte. Transplantation. 1995; 60(6):594-601. (Clone-specific: Immunohistochemistry). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Report a Site Issue
This form is intended to help us improve our website experience. For other support, please visit our Contact Us page.