Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?

.png)

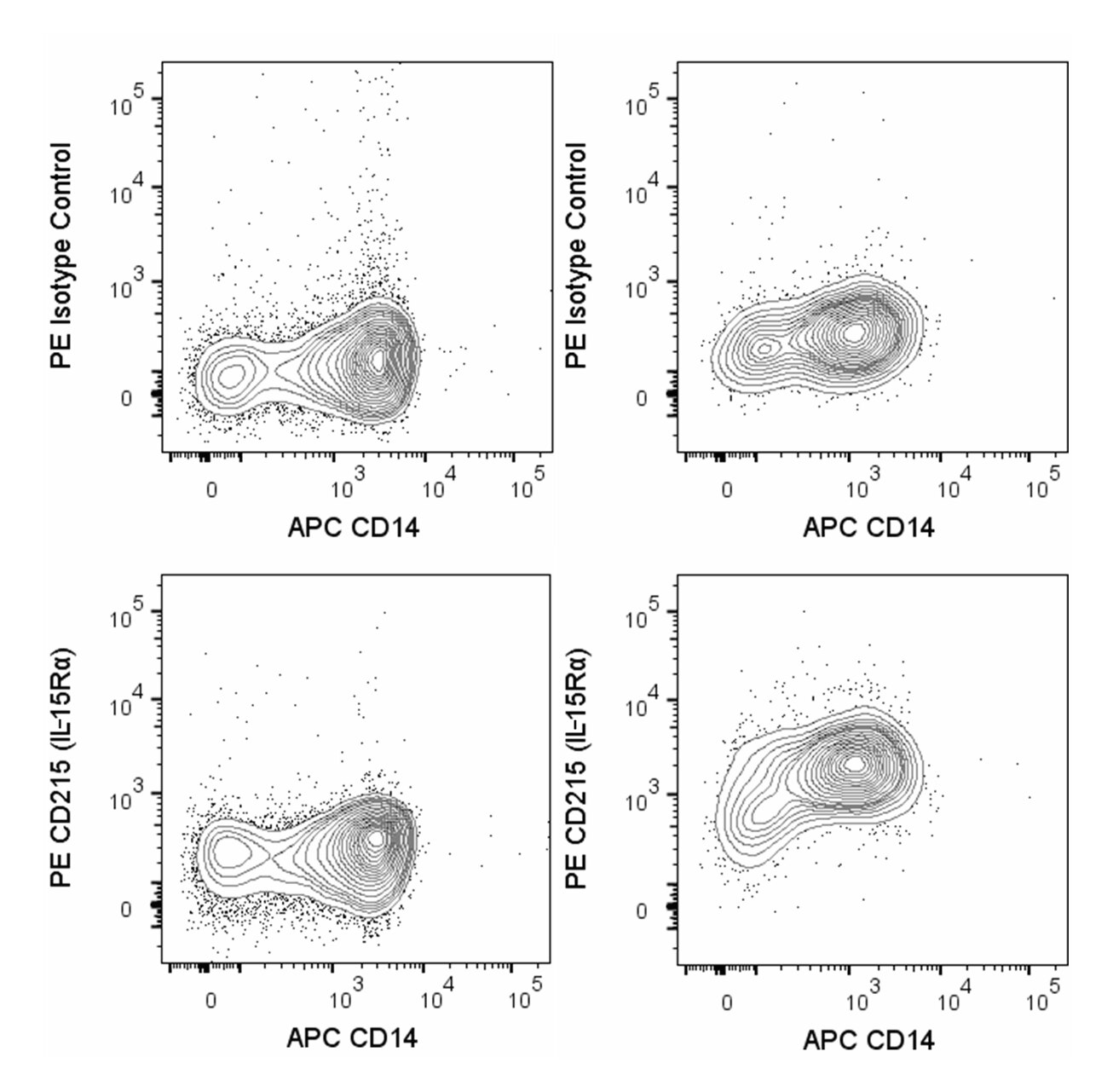
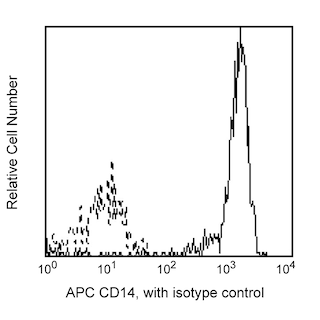
Multiparameter flow cytometric analysis of CD215 (IL15Rα) expression on human monocytes. Human peripheral blood mononuclear cells were cultured in complete tissue culture medium without (left column) or with (right column) lipopolysaccharide (LPS, 100 ng/ml) and Recombinant Human IFN-γ (Cat. No. 554617, 10 ng/ml) for 20 hours at 37°C. The cells were harvested, incubated with Human BD Fc Block™ (Cat. No. 564220), and then stained with APC Mouse Anti-Human CD14 (Cat. No. 555399) and either PE Mouse IgG2b, κ Isotype Control (Cat. No. 555058; top row) or PE Mouse Anti-Human CD215 (IL15Rα) antibody (Cat. No. 566589; bottom row). Two-color flow cytometric contour plots showing the correlated expression of CD215 (or Ig Isotype control staining) versus CD14 were derived from gated events with the forward and side light-scatter characteristics of viable monocytes. Flow cytometric analysis was performed using a BD LSRFortessaTM X-20 Flow Cytometer System. Data shown on this Technical Data Sheet are not lot specific.
.png)

BD Pharmingen™ PE Mouse Anti-Human CD215 (IL-15Rα)
.png)
Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
Product Notices
- This reagent has been pre-diluted for use at the recommended Volume per Test. We typically use 1 × 10^6 cells in a 100-µl experimental sample (a test).
- An isotype control should be used at the same concentration as the antibody of interest.
- Source of all serum proteins is from USDA inspected abattoirs located in the United States.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- For fluorochrome spectra and suitable instrument settings, please refer to our Multicolor Flow Cytometry web page at www.bdbiosciences.com/colors.
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
Companion Products
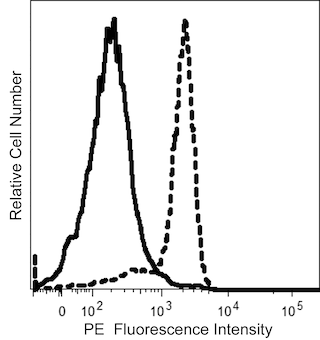


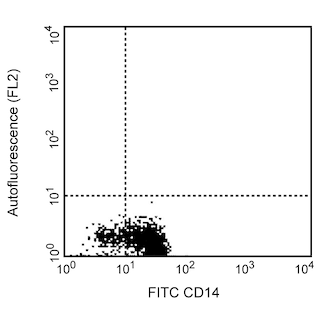
The JM7A4 monoclonal antibody specifically binds to CD215, which is also known as the IL-15 Receptor alpha subunit (IL-15R-alpha, IL-15Ra, or IL-15Rα). This type I transmembrane glycoprotein is encoded by IL15RA (Interleukin 15 receptor subunit alpha) and is variably expressed on macrophages, natural killer (NK) cells, T cells and B cells and by some nonlymphoid cells including fibroblasts. Although it can independently bind IL-15 with high affinity, it does not contain a signaling motif. CD215 (IL-15Rα) can present IL-15 in cis or trans fashion to the IL-2/15R beta (CD122) and IL-2R gamma (γc or CD132) receptor complex which can then transduce signals intracellularly. Several different CD215 (IL-15Rα) isoforms have been described that are produced by alternative splicing and may alter signal transduction responses to IL-15. A cleaved soluble form of CD215 known as sIL-15RA has also been reported which can bind and antagonize IL-15 activity. By binding to its heterotrimeric receptor, IL-15 plays crucial roles in innate immunity, eg, through the activation of NK cells and adaptive immunity, eg, in enhancing the survival of CD8+ memory T cells.
Development References (5)
-
Dubois S, Mariner J, Waldmann TA, Tagaya Y. IL-15Ralpha recycles and presents IL-15 In trans to neighboring cells.. Immunity. 2002; 17(5):537-47. (Immunogen: Flow cytometry). View Reference
-
Llinas L, Lazaro A, de Salort J, Matesanz-Isabel J, Sintes J, Engel P. Expression profiles of novel cell surface molecules on B-cell subsets and plasma cells as analyzed by flow cytometry. Immunol Lett. 2011; 134(2):113-121. (Clone-specific: Flow cytometry, Immunohistochemistry). View Reference
-
Matesanz-Isabel J, Sintes J, Llinas L, de Salort J, Lazaro A, Engel P. New B-cell CD molecules. Immunol Lett. 2011; 134(2):104-112. (Clone-specific: Flow cytometry). View Reference
-
Mortier E, Bernard J, Plet A, Jacques Y. Natural, proteolytic release of a soluble form of human IL-15 receptor alpha-chain that behaves as a specific, high affinity IL-15 antagonist.. J Immunol. 2004; 173(3):1681-8. (Biology). View Reference
-
Vámosi G, Bodnár A, Vereb G, et al. IL-2 and IL-15 receptor alpha-subunits are coexpressed in a supramolecular receptor cluster in lipid rafts of T cells.. Proc Natl Acad Sci USA. 2004; 101(30):11082-7. (Clone-specific: Immunofluorescence). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Report a Site Issue
This form is intended to help us improve our website experience. For other support, please visit our Contact Us page.