-
Your selected country is
Middle East / Africa
- Change country/language
Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?




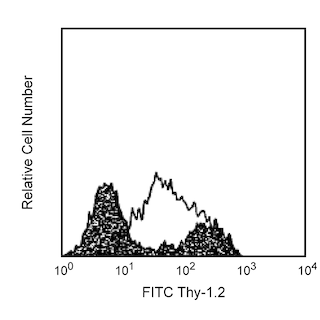
Flow cytometric analysis of CD25 expression on unstimulated and stimulated mouse splenic leucocytes. Left and Middle Plots: Mouse splenic leucocytes were preincubated with Purified Rat Anti-Mouse CD16/CD32 antibody (Mouse BD Fc Block™) (Cat. No. 553141/553142). The cells were then stained with FITC Rat Anti-Mouse CD4 antibody (Cat. No. 553047/553046/561835) and either BD Horizon™ BB700 Rat IgG1, κ Isotype Control (Cat. No. 566420; Left Plot) or BD Horizon BB700 Rat Anti-Mouse CD25 antibody (Cat. No. 566498/566499; Right Plot) at 0.5 µg/test. BD Pharmingen™ DAPI Solution (Cat. No. 564907) was added to cells right before analysis. Two-color flow cytometric dot plots showing the correlated expression of CD25 (or Ig Isotype control staining) versus CD4 were derived from DAPI negative-gated events with the forward and side light-scatter characteristics of viable leucocytes. Right Plot: Mouse splenic leucocytes were stimulated for 3 days with Concanavalin A (ConA). The cells were preincubated with Purified Rat Anti-Mouse CD16/CD32 antibody and then stained with either BD Horizon BB700 Rat IgG1, λ Isotype Control (dashed line histogram) or BD Horizon BB700 Rat Anti-Mouse CD25 antibody (solid line histogram) at 0.5 µg/test. The fluorescence histogram showing CD25 expression (or Ig Isotype control staining) was derived from DAPI negative-events with the forward and side light-scatter characteristics of viable lymphoblasts. Flow cytometric analysis was performed using a BD FACSCelesta™ Flow Cytometer System. Data shown on this Technical Data Sheet are not lot specific.


BD Horizon™ BB700 Rat Anti-Mouse CD25

Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
Recommended Assay Procedures
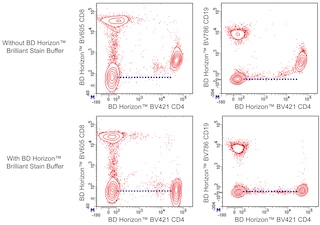
For optimal and reproducible results, BD Horizon Brilliant Stain Buffer should be used anytime two or more BD Horizon Brilliant dyes are used in the same experiment. Fluorescent dye interactions may cause staining artifacts which may affect data interpretation. The BD Horizon Brilliant Stain Buffer was designed to minimize these interactions. More information can be found in the Technical Data Sheet for the BD Horizon Brilliant Stain Buffer (Cat. No. 563794/566349) or the BD Horizon Brilliant Stain Buffer Plus (Cat. No. 566385).
When setting up compensation, it is recommended to compare spillover values obtained from cells and BD™ CompBeads to ensure that beads will provide sufficiently accurate spillover values.
For optimal results, it is recommended to perform two washes after staining with antibodies. Cells may be prepared, stained with antibodies and washed twice with wash buffer per established protocols for immunofluorescent staining, prior to acquisition on a flow cytometer. Performing fewer than the recommended wash steps may lead to increased spread of the negative population.
Product Notices
- Since applications vary, each investigator should titrate the reagent to obtain optimal results.
- An isotype control should be used at the same concentration as the antibody of interest.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- For fluorochrome spectra and suitable instrument settings, please refer to our Multicolor Flow Cytometry web page at www.bdbiosciences.com/colors.
- The manufacture, use, sale, offer for sale, or import of this product is subject to one or more patents or pending applications. This product, and only in the amount purchased by buyer, may be used solely for buyer’s own internal research, in a manner consistent with the accompanying product literature. No other right to use, sell or otherwise transfer (a) this product, or (b) its components is hereby granted expressly, by implication or by estoppel. Diagnostic uses require a separate license.
- BD Horizon Brilliant Stain Buffer is covered by one or more of the following US patents: 8,110,673; 8,158,444; 8,575,303; 8,354,239.
- BD Horizon Brilliant Blue 700 is covered by one or more of the following US patents: 8,455,613 and 8,575,303.
- Cy is a trademark of GE Healthcare.
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
Companion Products




The PC61 monoclonal antibody specifically binds to CD25, the low-affinity IL-2 Receptor α chain (IL-2Rα, p55) expressed on activated T and B lymphocytes from all mouse strains tested. IL-2Rα by itself is not a signaling receptor. However, it can combine with IL-2 Receptor β (CD122) and γc (CD132) chains to form high-affinity, signaling receptor complexes for IL-2. Resting T and B lymphocytes and resting and activated NK cells do not express IL-2Rα. CD25 is transiently expressed at a low level during normal B-cell development in the bone marrow on the CD45R/B220low TdT- sIg- Pre-B/Pre-B-II and CD45R/B220low TdT- sIgM+ sIgD- immature B stages, but not on the CD45R/B220low TdT+ sIg- Pro-B/Pre-B-I stage nor on CD45R/B220high TdT- sIgM+ sIgD+ mature B cells. It is expressed at a higher level during a very early stage of T-cell development in fetal and adult thymus. Peripheral CD25+CD4+ lymphocytes called regulatory T (Treg) cells are involved in the maintenance of self-tolerance. It has also been reported that dendritic cells express CD25, recognized by mAb 7D4. The PC61 antibody recognizes an epitope of CD25 which is distinct from the IL-2 binding site and from those recognized by mAbs 3C7 and 7D4. It blocks binding of IL-2 to CD25, presumably by inducing a conformational change in CD25.
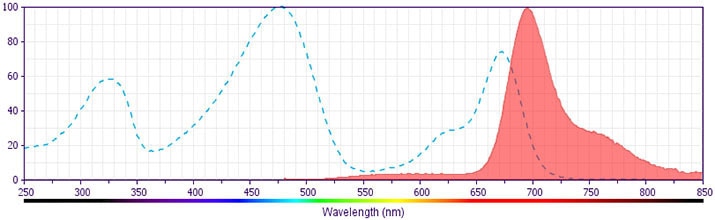
The antibody was conjugated to BD Horizon BB700, which is part of the BD Horizon Brilliant™ Blue family of dyes. It is a polymer-based tandem dye developed exclusively by BD Biosciences. With an excitation max of 485 nm and an emission max of 693 nm, BD Horizon BB700 can be excited by the 488 nm laser and detected in a standard PerCP-Cy™5.5 set (eg, 695/40-nm filter). This dye provides a much brighter alternative to PerCP-Cy5.5 with less cross laser excitation off the 405 nm and 355 nm lasers.
Development References (9)
-
Ceredig R, Lowenthal JW, Nabholz M, MacDonald HR. Expression of interleukin-2 receptors as a differentiation marker on intrathymic stem cells. Nature. 1985; 314(6006):98-100. (Clone-specific: Blocking, Immunohistochemistry). View Reference
-
Chen J, Ma A, Young F, Alt FW. IL-2 receptor alpha chain expression during early B lymphocyte differentiation. Int Immunol. 1994; 6(8):1265-1268. (Biology). View Reference
-
Ernst DN, Weigle WO, McQuitty DN, Rothermel AL, Hobbs MV. Stimulation of murine T cell subsets with anti-CD3 antibody. Age-related defects in the expression of early activation molecules. J Immunol. 1989; 142(5):1413-1421. (Clone-specific: Flow cytometry). View Reference
-
Garni-Wagner BA, Witte PL, Tutt MM, et al. Natural killer cells in the thymus. Studies in mice with severe combined immune deficiency. J Immunol. 1990; 144(3):796-803. (Biology). View Reference
-
Godfrey DI, Zlotnik A. Control points in early T-cell development. Immunol Today. 1993; 14(11):547-553. (Biology). View Reference
-
Lowenthal JW, Corthésy P, Tougne C, Lees R, MacDonald HR, Nabholz M. High and low affinity IL 2 receptors: analysis by IL 2 dissociation rate and reactivity with monoclonal anti-receptor antibody PC61. J Immunol. 1985; 135(6):3988-3994. (Immunogen: Bioassay, Blocking, Functional assay, Inhibition, Radioimmunoassay). View Reference
-
Lowenthal JW, Zubler RH, Nabholz M, MacDonald HR. Similarities between interleukin-2 receptor number and affinity on activated B and T lymphocytes. Nature. 1985; 315(6021):669-672. (Clone-specific: Blocking, Immunoprecipitation, Radioimmunoassay). View Reference
-
Moreau JL, Nabholz M, Diamantstein T, Malek T, Shevach E, Theze J. Monoclonal antibodies identify three epitope clusters on the mouse p55 subunit of the interleukin 2 receptor: relationship to the interleukin 2-binding site. Eur J Immunol. 1987; 17(7):929-935. (Clone-specific: Blocking). View Reference
-
Read S, Malmstrom V, Powrie F. Cytotoxic T lymphocyte-associated antigen 4 plays an essential role in the function of CD25(+)CD4(+) regulatory cells that control intestinal inflammation. J Exp Med. 2000; 192(2):295-302. (Biology). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Report a Site Issue
This form is intended to help us improve our website experience. For other support, please visit our Contact Us page.