Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?




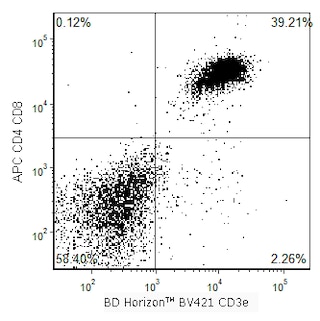
Multiparameter flow cytometric analysis of CD8b expression on mouse splenocytes. Mouse AKR splenic leucocytes were stained with BD Horizon™ BV421 Hamster Anti-Mouse CD3e antibody (Cat. No. 562600) and with either Alexa Fluor™ 647 Rat IgG2b, κ Isotype Control (Cat. No. 565378; Left Plot) or Alexa Fluor™ 647 Rat Anti-Mouse CD8b antibody (Cat. No. 567663; Right Plot) at 0.06 µg/test. BD Via-Probe™ Cell Viability 7-AAD Solution (Cat. No. 555815/555816) was added to cells right before analysis. A bivariate pseudocolor density plot showing the correlated expression of CD8b (or Ig Isotype control staining) versus CD3 was derived from gated events with the forward and side-light scatter characteristics of viable (7AA-D-negative) leucocytes. Flow cytometry and data analysis were performed using a BD LSRFortessa™ X-20 Cell Analyzer System and FlowJo™ software. Data shown on this Technical Data Sheet are not lot specific.


BD Pharmingen™ Alexa Fluor™ 647 Rat Anti-Mouse CD8b

Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
Recommended Assay Procedures
BD® CompBeads can be used as surrogates to assess fluorescence spillover (Compensation). When fluorochrome conjugated antibodies are bound to BD® CompBeads, they have spectral properties very similar to cells. However, for some fluorochromes there can be small differences in spectral emissions compared to cells, resulting in spillover values that differ when compared to biological controls. It is strongly recommended that when using a reagent for the first time, users compare the spillover on cells and BD® CompBeads to ensure that BD® CompBeads are appropriate for your specific cellular application.
Product Notices
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
- The Alexa Fluor®, Pacific Blue™, and Cascade Blue® dye antibody conjugates in this product are sold under license from Molecular Probes, Inc. for research use only, excluding use in combination with microarrays, or as analyte specific reagents. The Alexa Fluor® dyes (except for Alexa Fluor® 430), Pacific Blue™ dye, and Cascade Blue® dye are covered by pending and issued patents.
- Alexa Fluor® 647 fluorochrome emission is collected at the same instrument settings as for allophycocyanin (APC).
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- Since applications vary, each investigator should titrate the reagent to obtain optimal results.
- For fluorochrome spectra and suitable instrument settings, please refer to our Multicolor Flow Cytometry web page at www.bdbiosciences.com/colors.
- An isotype control should be used at the same concentration as the antibody of interest.
- This product is provided under an intellectual property license between Life Technologies Corporation and BD Businesses. The purchase of this product conveys to the buyer the non-transferable right to use the purchased amount of the product and components of the product in research conducted by the buyer (whether the buyer is an academic or for-profit entity). The buyer cannot sell or otherwise transfer (a) this product (b) its components or (c) materials made using this product or its components to a third party or otherwise use this product or its components or materials made using this product or its components for Commercial Purposes. Commercial Purposes means any activity by a party for consideration and may include, but is not limited to: (1) use of the product or its components in manufacturing; (2) use of the product or its components to provide a service, information, or data; (3) use of the product or its components for therapeutic, diagnostic or prophylactic purposes; or (4) resale of the product or its components, whether or not such product or its components are resold for use in research. For information on purchasing a license to this product for any other use, contact Life Technologies Corporation, Cell Analysis Business Unit Business Development, 29851 Willow Creek Road, Eugene, OR 97402, USA, Tel: (541) 465-8300. Fax: (541) 335-0504.
- Please refer to http://regdocs.bd.com to access safety data sheets (SDS).
- Alexa Fluor™ is a trademark of Life Technologies Corporation.
Companion Products




The H35-17.2 monoclonal antibody specifically binds to both alloantigeneic forms of the β chain of the CD8 differentiation antigen (Ly-3 or Lyt- 3). The CD8 α and α' chains (CD8a) form heterodimers with the CD8 β chain (CD8b, Ly-3, or Lyt-3) on the surface of most thymocytes. A subpopulation of mature T lymphocytes (i.e., MHC class I-restricted T cells, including most T suppressor/cytotoxic cells) expresses almost exclusively the CD8 αβ heterodimer (the α' chain is absent). Subsets of γδ TCR-bearing T cells, intestinal intraepithelial lymphocytes, and dendritic cells express CD8a without CD8b. It has been suggested that the expression of the CD8a/CD8b heterodimer is restricted to T lymphocytes which matured in the thymus or in an extrathymic environment that had been influenced by thymus- initiated neuroendocrine signals. CD8 is an antigen coreceptor on the T-cell surface which interacts with MHC class I molecules on antigen-presenting cells. It participates in T-cell activation through its association with the T-cell receptor complex and protein tyrosine kinase lck (p56lck). The H35-17.2 mAb blocks T-cell-mediated cytolysis of allogeneic lymphoma cells.
Development References (9)
-
Golstein P, Goridis C, Schmitt-Verhulst AM . Lymphoid cell surface interaction structures detected using cytolysis-inhibiting monoclonal antibodies. Immunol Rev. 1982; 68:5-42. (Biology). View Reference
-
Golstein P, Pierres M, Schmitt-Verhulst AM, et al. Functional relationships of lymphocyte membrane structures probed with cytolysis and/or proliferation-inhibiting H35-27.9 and H35-89.9 monoclonal antibodies.. Adv Exp Med Biol. 1982; 146:487-503. (Clone-specific: Functional assay, Inhibition). View Reference
-
Lefrancois L. Phenotypic complexity of intraepithelial lymphocytes of the small intestine. J Immunol. 1991; 147(6):1746-1751. (Clone-specific: Flow cytometry). View Reference
-
MacDonald HR, Schreyer M, Howe RC, Bron C. Selective expression of CD8 alpha (Ly-2) subunit on activated thymic gamma/delta cells. Eur J Immunol. 1990; 20(4):927-930. (Clone-specific: Flow cytometry). View Reference
-
Nakayama K, Nakayama K, Negishi I, et al. Requirement for CD8 beta chain in positive selection of CD8-lineage T cells. Science. 1994; 263(5150):1131-1133. (Biology). View Reference
-
Pierres A, Naquet P, Van Agthoven A, et al. A rat anti-mouse T4 monoclonal antibody (H129.19) inhibits the proliferation of Ia-reactive T cell clones and delineates two phenotypically distinct (T4+, Lyt-2,3-, and T4-, Lyt-2,3+) subsets among anti-Ia cytolytic T cell clones. J Immunol. 1984; 132(6):2775-2782. (Clone-specific: Flow cytometry). View Reference
-
Pierres M, Goridis C, Golstein P. Inhibition of murine T cell-mediated cytolysis and T cell proliferation by a rat monoclonal antibody immunoprecipitating two lymphoid cell surface polypeptides of 94 000 and 180 000 molecular weight.. Eur J Immunol. 1982; 12(1):60-9. (Immunogen: Cytotoxicity, Radioimmunoassay). View Reference
-
Vremec D, Zorbas M, Scollay R, et al. The surface phenotype of dendritic cells purified from mouse thymus and spleen: investigation of the CD8 expression by a subpopulation of dendritic cells. J Exp Med. 1992; 176(1):47-58. (Biology). View Reference
-
Zamoyska R. The CD8 coreceptor revisited: one chain good, two chains better. Immunity. 1994; 1(4):243-246. (Biology). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Report a Site Issue
This form is intended to help us improve our website experience. For other support, please visit our Contact Us page.