Old Browser
This page has been recently translated and is available in French now.
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?


.png)

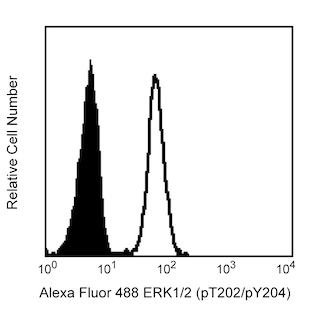
Flow cytometric analysis of ERK1/2 (pT202/pY204). Human peripheral blood mononulcear cells (PBMC) were either left unstimulated (unshaded) or stimulated (shaded) with 40 nM PMA for 10 minutes at 37°C. Cells were fixed with BD Cytofix™ buffer (Cat. No. 554655) for 10 minutes at 37°C and then permeabilized by adding BD Phosflow™ Perm Buffer III (Cat. No. 558050) for 30 minutes on ice. Cells were then washed twice in BD Pharmingen™ Stain Buffer (Cat. No. 554656) and stained with the Alexa Fluor® 488 mouse anti-ERK1/2 (pT202/pY204) antibody. Cells were analyzed on a BD FACSCalibur™ flow cytometry instrument. For intracellular staining of human whole blood, BD Phosflow™ Lyse/Fix buffer (Cat. No. 558049) may be used for fixation.
.png)

BD™ Phosflow Alexa Fluor® 488 Mouse Anti-ERK1/2 (pT202/pY204)
.png)
Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
Recommended Assay Procedures
For more information investigators are encouraged to reference http://www.bdbiosciences.com/research/ics/resources/index.jsp
Product Notices
- This reagent has been pre-diluted for use at the recommended Volume per Test. We typically use 1 × 10^6 cells in a 100-µl experimental sample (a test).
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
- Alexa Fluor® 488 fluorochrome emission is collected at the same instrument settings as for fluorescein isothiocyanate (FITC).
- The Alexa Fluor®, Pacific Blue™, and Cascade Blue® dye antibody conjugates in this product are sold under license from Molecular Probes, Inc. for research use only, excluding use in combination with microarrays, or as analyte specific reagents. The Alexa Fluor® dyes (except for Alexa Fluor® 430), Pacific Blue™ dye, and Cascade Blue® dye are covered by pending and issued patents.
- Alexa Fluor® is a registered trademark of Molecular Probes, Inc., Eugene, OR.
- For fluorochrome spectra and suitable instrument settings, please refer to our Multicolor Flow Cytometry web page at www.bdbiosciences.com/colors.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
- Source of all serum proteins is from USDA inspected abattoirs located in the United States.
- Species cross-reactivity detected in product development may not have been confirmed on every format and/or application.
Companion Products



The members of the Mitogen-Activated Protein Kinase (MAPK) family are components of a key signal transduction cascade that links events at the cell surface to responses in the nucleus. The signaling cascade is found in species as varied as yeast and humans, with many of the proteins being well conserved. In mammals the most widely studied members of the cascade are the Extracellular signal-Regulated Kinases, ERK1 (p44 MAPK) and ERK2 (p42 MAPK). ERK1 and ERK2 share 85% homology and are activated by extracellular signals such as growth factors, hormones, and phorbol esters. Activation occurs through a series of phosphorylations by kinases activating other kinases and eventually leading to phosphorylation of the ERKs. Growth factor stimulation leads to activation of Ras and Raf, leading to phosphorylation of MEK1 (MAPK/ERK kinase) which, in turn, activates the ERKs via dual phosphorylation. Once activated, the ERKs phosphorylate other cytoplasmic signalling molecules, cell-surface receptors, microtubule-associated proteins, and transcription factors in the nucleus. Thus, the active ERK has myriad downstream effectors that implicate it in the control of cell proliferation and differentiation, as well as regulation of the cytoskeleton. Furthermore, studies have shown that elevated ERK activity is associated with some cancers.
The 20A monoclonal antibody recognizes the phosphorylated threonine 202 and tyrosine 204 (pT202/pY204) of human ERK1 and pT184/pY186 of human ERK2. The orthologous phosphorylation sites in murine ERK1 and ERK2 are T203/Y205 and T183/Y185.
Development References (7)
-
Boulton TG, Cobb MH. Identification of multiple extracellular signal-regulated kinases (ERKs) with antipeptide antibodies. Cell Regul. 1991; 2(5):357-371. (Biology). View Reference
-
Clark EA, Hynes RO. Ras activation is necessary for integrin-mediated activation of extracellular signal-regulated kinase 2 and cytosolic phospholipase A2 but not for cytoskeletal organization. J Biol Chem. 1996; 271(25):14814-14818. (Biology). View Reference
-
Irish JM, Czerwinski DK, Nolan GP, Levy R. Altered B-cell receptor signaling kinetics distinguish human follicular lymphoma B cells from tumor-infiltrating nonmalignant B cells. Blood. 2006; 108(9):3135-3142. (Clone-specific: Flow cytometry). View Reference
-
Irish JM, Czerwinski DK, Nolan GP, Levy R. Kinetics of B cell receptor signaling in human B cell subsets mapped by phosphospecific flow cytometry. J Immunol. 2006; 177(3):1581-1589. (Clone-specific: Flow cytometry). View Reference
-
Krutzik PO, Nolan GP. Fluorescent cell barcoding in flow cytometry allows high-throughput drug screening and signaling profiling. Nat Methods. 2006; 3(5):361-368. (Clone-specific: Flow cytometry). View Reference
-
Krutzik PO, Nolan GP. Intracellular phospho-protein staining techniques for flow cytometry: monitoring single cell signaling events. Cytometry A. 2003; 55(2):61-70. (Clone-specific: Flow cytometry). View Reference
-
Sivaraman VS, Wang H, Nuovo GJ, Malbon CC. Hyperexpression of mitogen-activated protein kinase in human breast cancer. J Clin Invest. 1997; 99(7):1478-1483. (Biology). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Report a Site Issue
This form is intended to help us improve our website experience. For other support, please visit our Contact Us page.