Old Browser
Looks like you're visiting us from {countryName}.
Would you like to stay on the current country site or be switched to your country?
BD™ DimerX DimerX I: Recombinant Soluble Dimeric Human CD1d:Ig Fusion Protein
Clone hCD1d/IgG1
(RUO)
Schematic representation of the CD1d:Ig dimeric protein


BD Pharmingen™ DimerX I: Recombinant Soluble Dimeric Human CD1d:Ig Fusion Protein

Regulatory Status Legend
Any use of products other than the permitted use without the express written authorization of Becton, Dickinson and Company is strictly prohibited.
Preparation And Storage
The human CD1d:Ig fusion protein was expressed together with human β2M in the mouse plasmacytoma cell line, J558L (ATCC TIB-6). The CD1d and β2M polypeptide chains are associated noncovalently as a consequence of their coexpression within J558L cells. The CD1d:Ig fusion protein was purified from tissue culture supernatant by affinity chromatography.The purity of the preparation was confirmed by SDS-PAGE. Additionally, human β2M was added to the reagent. The human CD1d:Ig is tested by ELISA to verify the fusion protein.
Recommended Assay Procedures
It is necessary to load the CD1d portions of the dimeric protein with a relevant antigen of interest prior to immunofluorescent staining of NKT cells. CD1d:Ig complexes are effectively loaded by incubation with excess relevant (specific) or irrelevant (control) antigens (see Protocol 1). Antigen-loaded CD1d:Ig may be used for immunofluorescent staining (see Protocol 2). Since applications vary, each investigator must determine dilutions appropriate for individual use.
Protocol 1: Antigen Loading of CD1d:Ig Dimeric Protein
Several antigen-loading protocols have been described. The method recommended at BD Biosciences Pharmingen involves passive loading of excess antigen in solution with CD1d:Ig protein. We have found that passive loading works particularly well in the case of high-affinity antigens. For lower-affinity antigens, an increase in the molar ratio of antigen to dimer protein may improve loading, as determined by flow cytometric analysis based on results for other BD Pharmingen DimerX products. It is suggested that for each antigen, parameters such as the dose of CD1d:Ig per million cells, molar ratio of antigen to CD1d:Ig, and antigen-loading time be determined empirically by the investigator.
Antigen preparation and loading:
1. The molecular weight (MW) of an antigen of interest will need to be determined. The MW of a-GalCer is 858 daltons.
2. Mix CD1d:Ig protein with specific or control antigen at 10, 20, or 40 molar (M) excess.
The following calculation, using α-GalCer as an example, may be used:
Dg = Molecular Weight of antigen: e.g., 858 daltons.
DCD1d = Molecular Weight of CD1d:Ig = 250,000 daltons.
R = desired excess molar ratio, e.g., 40.
Mg = micrograms (µg) antigen of interest.
MCD1d = micrograms (µg) CD1d:Ig in the reaction. A typical amount of antigen-loaded CD1d:Ig to use for flow cytometry staining is 0.25
to 4 µg/million cells (test).
Mg = MCD1d x R x Dg = 4 µg x 40 x 858 d = 0.55 µg
DCD1d 250,000 d
Therefore, one would add 0.55 µg of antigen and 4 µg of CD1d:Ig in solution for the optimal antigen loading of CD1d:Ig.
3. Mix antigen and CD1d :Ig together in PBS, pH 7.2, incubate at 37°C overnight. The antigen-loaded CD1d:Ig can be stored at 4°C for up to 1 week.
*NOTE: The rights to α-GalCer are owned by Kirin Brewery. The α-GalCer molecule and its derivatives are covered by US Patent No. 5,936,076. There is no implied license hereunder for the use of α-GalCer.
Protocol 2: Immunofluorescent Staining Protocol
1. Resuspend PBMC's or target cells in FACS staining buffer [e.g., BD Pharmingen™ Stain Buffer with BSA, Cat. no. 554657], at a concentration of approximately 10e6 cells per 50 µl. Add ~1 x 10e6 cells per staining tube (eg, 12 x 75 mm tube, BD Falcon™ Cat. no. 352008).
2. Prepare antigen-loaded CD1d:Ig protein staining cocktail by mixing 0.25 - 4 µg of antigen-loaded CD1d:Ig protein/test with 0.25 - 4 µg of PE-conjugated A85-1 mAb (anti-mouse IgG1, Cat. No. 550083)/test at a ratio of 1:1 or 1:2 of dimer: A85-1 mAb. Incubate the mixture for 60 minutes at room temperature, protect from exposure to light.
3. Add 1 -2 µg of purified mouse IgG1 isotype control mAb A111-3 (Cat. No. 553485)/test to the staining cocktail (see Step 2 above). Incubate the staining cocktail for 30 minutes at RT, protect from exposure to light.
4. Prepare purified polyclonal human IgG at approximately 0.2 mg/ml in phosphate buffered saline (PBS), pH 7.2.
5. Add 10 µl (2 µg) of human IgG stock per tube to block non-specific binding of DimerX I or antibody reagents to surface Fc receptors. Incubate 10 minutes at room temperature.
6. Add 50 µl FACS buffer containing the optimal per test amount of the staining cocktail, plus any other cell-surface marker-specific antibodies to be used to each sample.
7. Wash cells 1x with 2 ml FACS buffer, centrifuge for 5 minutes at 250 x g, and aspirate supernatant. Resuspend in FACS buffer and analyze by flow cytometry.
Protocol 3: Alternative: Immunofluorescent Staining Protocol
1. Resuspend PBMCs or target cells in FACS staining buffer [eg, DPBS, 1% FCS, 0.09% NaN3 or BD Pharmingen™ Stain Buffer (FBS), Cat. No. 554656] at a concentration of approximately 10e6 cells per 50 µl. Add ~1 x 10e6 cells per staining tube (e.g., 12 x 75 mm tube, BD Falcon™ Cat. no. 352008).
2. Prepare purified polyclonal human IgG at approximately 0.2 mg/ml in phosphate buffered saline (PBS), pH 7.2.
3. Add 10 µl (2 µg) of human IgG stock per tube to block non-specific binding of DimerX I or antibody reagents to surface Fc receptors. Incubate 10 minutes at room temperature.
4. Add 0.25 to 4 µg of antigen-loaded CD1d:Ig protein to cell suspension. Incubate 60 minutes at 4°C.
5. Wash cells 1x with 2 ml FACS buffer, centrifuge for 5 minutes at 250 x g, and discard supernatant.
6. Again add 10 µl (2 µg) of purified polyclonal human IgG per sample.
7. Resuspend cells in 100 µl FACS buffer containing appropriately diluted fluorescent secondary reagent. We typically use PE-conjugated A85-1 mAb (anti-mouse IgG1, Cat. No. 550083). Incubate 30 - 60 minutes at 4°C.
8. Wash cells 1x with 2 ml FACS buffer, centrifuge for 5 minutes at 250 x g, and discard supernatant.
9. Repeat Steps 7 and 8 for labeling of cell-surface markers with the appropriate fluorochrome-conjugated antibody (avoid antibodies with mouse IgG1 isotypes to reduce background).
10. Resuspend cell pellet in approximately 0.5 ml staining buffer in a tube appropriate for the flow cytometer and analyze.
Product Notices
- Since applications vary, each investigator should titrate the reagent to obtain optimal results.
- Please refer to www.bdbiosciences.com/us/s/resources for technical protocols.
- Caution: Sodium azide yields highly toxic hydrazoic acid under acidic conditions. Dilute azide compounds in running water before discarding to avoid accumulation of potentially explosive deposits in plumbing.
Companion Products

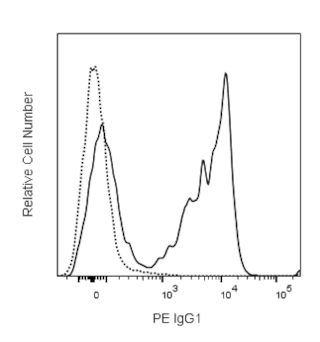
The CD1d:Ig fusion protein consists of the extracellular major histocompatibility complex (MHC) class I-like domains of the human CD1d molecule fused with the VH regions of mouse IgG1. Like MHC class I molecules, the CD1d noncovalently associates with β2 Microglobulin (β2M). For this reason, BD Pharmingen™ DimerX consists of recombinant CD1d:Ig and human β2M. Recombinant CD1d molecules, like the DimerX fusion protein, are useful for studying Natural Killer T (NKT)-cell function by immunofluorescent staining and flow cytometric analysis of antigen-specific NKT cells.
The human CD1D1 gene encodes a non-polymorphic cell-surface protein that plays a role in antigen presentation to CD1d-restricted NKT cells. Like the MHC class I molecules, CD1d associates noncovalently with β2M and is capable of binding and presenting lipid antigens. While the natural ligand for CD1d is presently unknown, it is well documented that CD1d can bind and present the glycolipid, α-galactosyl ceramide (α-GalCer*), a glycosphingolipid from the marine sponge. Glycosylphosphatidylinositol (GPI) has also been identified as a major CD1d-associated component. Antigenic glycolipids such as α-GalCer associated with the CD1d molecule are presented and specifically recognized by NKT cells expressing a highly conserved TCR, Vα24JαQ, paired with Vβ 11. The human CD1d-α-GalCer complex has been shown to stain a subpopulation of Vα 24+ cells. The human CD1d:Ig dimer presenting the α-GalCer molecule has been shown to stain a higher percentage of Vα 14Ja281+Vβ8.2+ NKT cells in the mouse than Vα 14+Vβ 7+ NKT cells. However, the percentage of mouse NKT cells stained by the human CD1d:Ig-α-GalCer complex was significantly lower than the percentage stained by mouse CD1d:Ig-α-GalCer. The function and lineage marker expression of NKT cells and their interaction with CD1d has been recently reviewed.
Development References (7)
-
Godfrey DI, Hammond KJ, Poulton LD, Smyth MJ, Baxter AG. NKT cells: facts, functions and fallacies. Immunol Today. 2000; 21(11):573-583. (Biology). View Reference
-
Karadimitris A, Gadola S, Altamirano M, et al. Human CD1d-glycolipid tetramers generated by in vitro oxidative refolding chromatography. Proc Natl Acad Sci U S A. 2001; 98(6):3294-3298. (Biology). View Reference
-
Lee PT, Benlagha K, Teyton L, Bendelac A. Distinct functional lineages of human V(alpha)24 natural killer T cells. J Exp Med. 2002; 195(5):637-641. (Biology). View Reference
-
Naidenko OV, Maher JK, Ernst WA, Sakai T, Modlin RL, Kronenberg M. Binding and antigen presentation of ceramide-containing glycolipids by soluble mouse and human CD1d molecules. J Exp Med. 1999; 190(8):1069-1080. (Biology). View Reference
-
Schneck JP, Slansky JE, O'Herrin SM, Greten TF . Monitoring antigen-specific T cells using MHC-Ig dimers. In: Coligan J, Kruisbeek D, Margulies EM, Shevach EM, Strober W, ed. Current Protocols in Immunology. New York: John Wiley & Sons, Inc; 2000:17.2.1-17.2.17.
-
Schumann J, Voyle RB, Wei BY, MacDonald HR. Cutting edge: influence of the TCR V beta domain on the avidity of CD1d:alpha-galactosylceramide binding by invariant V alpha 14 NKT cells. J Immunol. 2003; 170(12):5815-5819. (Biology). View Reference
-
Sidobre S, Kronenberg M. CD1 tetramers: a powerful tool for the analysis of glycolipid-reactive T cells. J Immunol Methods. 2002; 268(1):107-121. (Biology). View Reference
Please refer to Support Documents for Quality Certificates
Global - Refer to manufacturer's instructions for use and related User Manuals and Technical data sheets before using this products as described
Comparisons, where applicable, are made against older BD Technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
For Research Use Only. Not for use in diagnostic or therapeutic procedures.
Refer to manufacturer's instructions for use and related User Manuals and Technical Data Sheets before using this product as described.
Comparisons, where applicable, are made against older BD technology, manual methods or are general performance claims. Comparisons are not made against non-BD technologies, unless otherwise noted.
Report a Site Issue
This form is intended to help us improve our website experience. For other support, please visit our Contact Us page.